Bias in Real-world Endpoints when Conducting External Control Arm Analyses
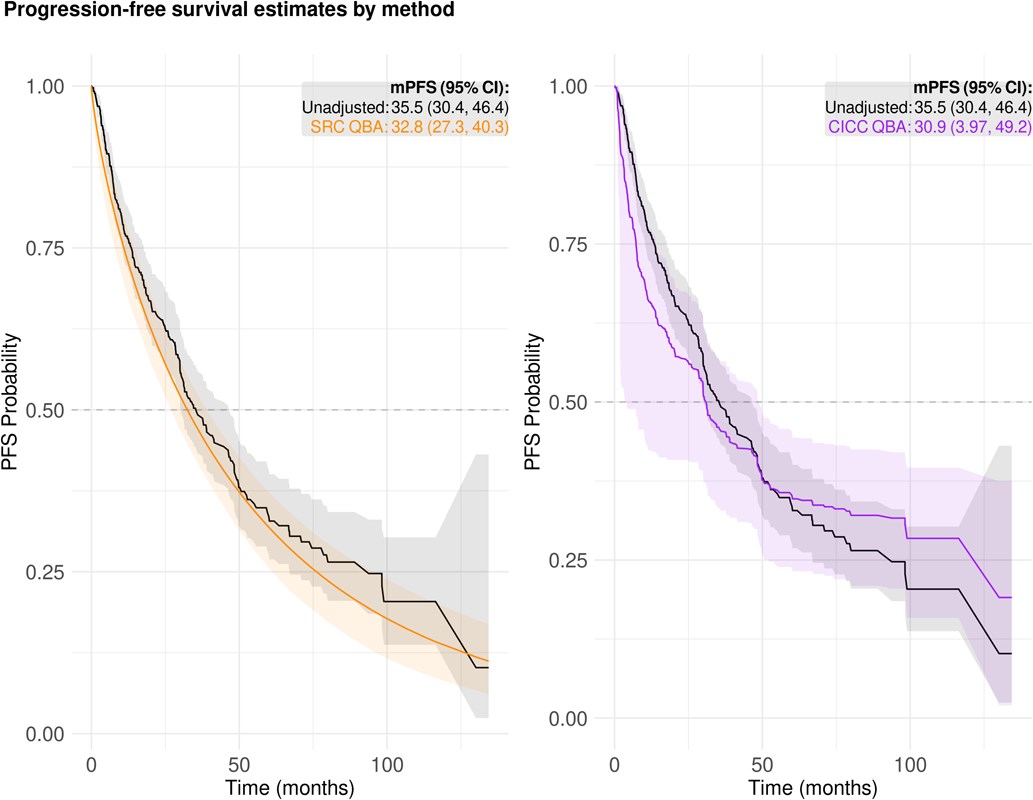
When augmenting single-arm trials with real-world data for external control arm analyses, biases may arise due to differences in how and when patients’ disease progression is assessed between the two settings. Funded by an FDA U01 methods research grant, I’ve developed methods and simulation tools to quantify the impact of these biases, calibrate real-world endpoints to be more trial-like, and conduct Quantitative Bias Analyses to contextualize ECA findings in the presence of endpoint measurement error.