Extending Inferences from Trials to Target Populations
Background
Randomized trials are often considered the gold standard for estimating causal effects and evaluating efficacy of new therapeutics. However, patients recruited for clinical trials are often not representative of the target populations of interest, and thus, findings from trials may not generalize or transport from the recruited sample population to broader populations.
Generalizability refers to whether inferences extend from the source study population to the target population from which it was sampled. Transportability refers to whether inferences extend from the source study population to a different population (e.g., one may wish to transport findings from a trial conducted in the US to a population of patients in Europe).
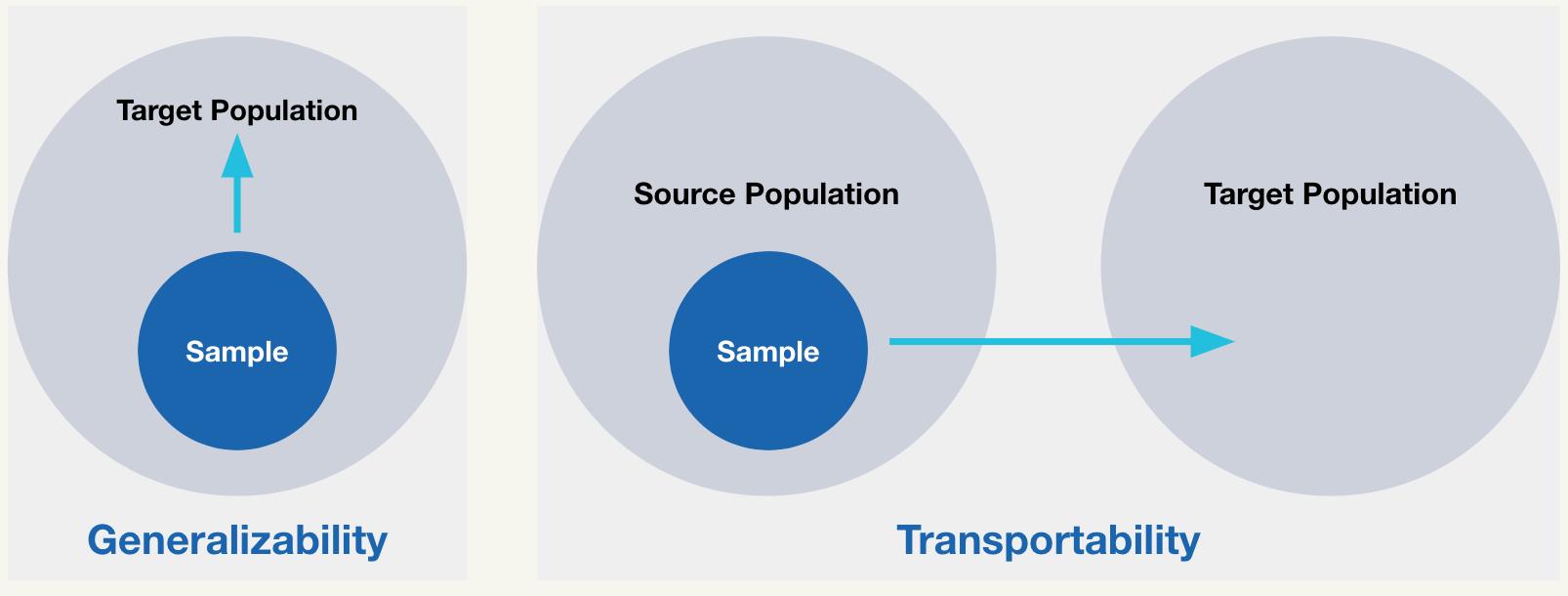
Both generalizability and transportability depend on representativeness, or the similarity between the trial participants and the target population with respect to key baseline factors. More specifically, without representativeness on characteristics that moderate the treatment effect, then trial findings may not extend to the target population of interest.
Key Findings and Takeaways
While it may not be feasible to always recruit trial participants that reflect the intended target populations,
- Evaluating representativeness on key factors that modify the treatment effect can inform how well trial findings generalize or transport to target populations of interest.
- Statistical methods, such as propensity score-type weighting or doubly-robust outcome modeling, can be applied to extend trial inferences to broader populations, using external population data as a reference frame.
- Finding suitable target population data can be challenging in practice, and may require further statistical adjustments when extending inferences (e.g., when using a nationally representative complex health survey).
Relevant Papers
Vuong, Q., Metcalfe, R.K., Ling, A., Ackerman, B., Inoue, K., Park, J.J.H. (2025). Systematic review of applied transportability and generalizability analyses: A landscape analysis. Annals of Epidemiology.
Gerke, T., Ackerman, B., Long, L., Baxi, S., Miksad, R., Adamson, B., et al. (2021). Representativeness of real world data: a framework for assessing oncology EHR-derived data. SSRN.
Ackerman, B., Lesko, C.R., Siddique, J., Susukida, R., Stuart, E.A. (2020). Generalizing randomized trial findings to a target population using complex survey population data. Statistics In Medicine.
Lesko, C.R., Ackerman, B., Webster-Clark, M., Edwards, J.K. (2020). Target validity: bringing treatment of external validity in line with internal validity. Current Epidemiology Reports.
Ackerman, B., Schmid, I., Rudolph, K.E., Seamans, M.J., Susukida, R., Mojtabai, R., Stuart, E.A. (2019). Implementing statistical methods for generalizing randomized trial findings to a target population. Addictive Behaviors.
Nguyen, T.Q., Ackerman, B., Schmid, I., Cole, S., Stuart, E.A. (2018). Sensitivity analyses for effect modifiers not observed in the target population when generalizing treatment effects from a randomized controlled trial: Assumptions, models, effect scales, data scenarios, and implementation details. PLoS One.